Abstract
Acute myeloid leukemia (AML) is an aggressive hematologic malignancy with poor prognosis. While most patients achieve initial remission following standard chemotherapy, the majority will relapse and ultimately die. Poor outcomes in AML are tied to the properties of leukemia stem cells (LSC), which drive therapy resistance and relapse. For successful therapeutic targeting, it is critical to understand the pathways that operate in LSC function and survival and how they are impacted by drug activity.
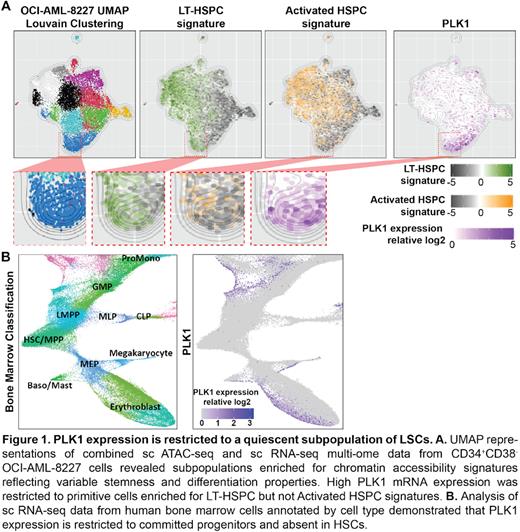
We carried out an innovative stemness-focused high-throughput drug screen employing a hierarchical AML cell model and a Nanostring-based LSC signature assay to probe for genes and pathways that impact LSC function. All 4 small molecule inhibitors of Polo-like Kinase 1 (PLK1) included in the compound library were among the top hits. PLK1 inhibitor treatment selectively targeted the CD34⁺CD38⁻ fraction of OCI-AML-8227 cells, which is enriched for quiescent LSCs. PLK1 is an important cell cycle regulator; although there are reports for cell cycle-independent roles, they have not been systematically characterized. Importantly, the role of PLK1 has not been previously studied in the context of cancer stem cells. To investigate the role of PLK1 in LSCs, we performed single-cell (sc) multi-omics incorporating both scRNAseq and scATACseq on the CD34⁺CD38⁻ compartment of OCI-AML-8227 cells. Enrichment analysis of previously-identified chromatin accessibility signatures of primitive long-term hematopoietic stem cells (LT-HSC) and progenitors was used to identify cell clusters with distinct stemness and differentiation properties. Interestingly, PLK1 mRNA expression in OCI-AML-8227 was restricted to cells enriched for the LT-HSC epigenetic signature (Fig 1A). In healthy human bone marrow, PLK1 expression was restricted to committed progenitors and not detected in HSCs (Fig 1B). These data strongly suggest a previously unexplored role of PLK1 in regulating stemness that may be specific LSCs and not normal HSCs.
To determine the effects of PLK1 against LSCs in vivo, we carried out PLK1 inhibitor treatment using the specific PLK1 inhibitor volasertib in a cohort of 9 primary AML samples in PDX models. Volasertib treatment significantly reduced leukemic burden in 7 of 9 samples, all of which had a high LSC17 score (associated with poor prognosis and standard chemotherapy response). To quantify effects on LSCs, we serially transplanted 2 samples at limiting doses into untreated secondary recipients and demonstrated a 5- to 25-fold reduction in LSC frequency following volasertib treatment, indicating that functional LSCs were targeted by PLK1 inhibition.
Together, these data strongly suggest that PLK1 impacts LSC function and stemness, and that PLK1 inhibitors are acting through an uncharacterized cell-cycle independent mechanism to target quiescent LSCs. To discover novel PLK1 signaling pathways, we carried out proximity-dependent biotinylation (PDB) to characterize the PLK1 interactome in serum-starved cells. Preliminary analysis has revealed a number of previously uncharacterized PLK1 interactors, including components of autophagy and apoptosis machinery, that are currently being validated in the hematopoietic cell context. Our study highlights PLK1 as a specific vulnerability of LSCs, and furthers the understanding of malignant PLK1 signaling in non-cell cycle related processes. Further insights into the underlying mechanisms will enable the development of effective therapies to target these pathways and improve patient outcomes in AML.
Disclosures
Dick:Graphite Bio: Membership on an entity's Board of Directors or advisory committees; Trillium Therapeutics/Pfizer: Patents & Royalties: patent licencing; Celgene/BMS: Research Funding. Wang:Trillium Therapeutics/Pfizer: Patents & Royalties: patent licensing.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal